What is 510(k)?
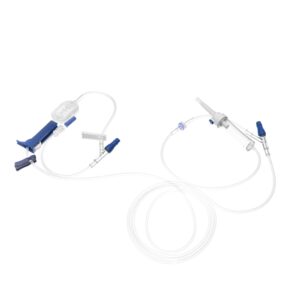
BQ Plus Medical Co., Ltd., a professional medical device manufacturer focused on contract manufacturing of IV Administration and Extension Set, announced today that it has received 510(k) clearance for IV Administration Set and Extension Set from the U.S. Food and Drug Administration (FDA).